Most rhinologic procedures, particularly endoscopic sinonasal procedures, are liable to produce aerosols. The severe acute respiratory syndrome coronavirus 2 (SARS-Co V-2) transmits via respiratory droplets, but the degree of its spread through airborne routes by aerosol is unclear.
ObjectiveThe aim of this article is to counsel rhinologists on how to modify their conventional practice during the COVID-19 pandemic by prioritising the need of procedures, identifying aerosol- generating procedures and using precise personal protection equipment for various endonasal procedures.
MethodsWe did a review of articles indexed for MEDLINE on PubMed, ENT Cochrane, DOAJ and Web of Science databases using the keywords nasal endoscopy, SARS‐CoV‐2, COVID‐19, aerosol generating medical procedures and rhinology to formulate guidelines for the safety of healthcare workers.
ResultsThe review included evidence from 28 articles from the otorhinolaryngology, surgery, infectious disease, head and neck surgery and cancer biology literature. We have provided recommendations and relevant information for rhinologists during the COVID-19 pandemic, based on the available studies and data, to warrant high-quality patient care and requisite levels of infection prevention during rhinology procedures.
ConclusionIn rhinology, marked care is advised during nasal packing, electrocauterisation and use of high-speed rotating devices in potentially infected tissue as they are considerable aerosol- producing procedures. The choice of personal protective equipment is based on the risk of exposure and possible modes of aerosol generation.
Reducing nosocomial infection is a challenge for all healthcare systems. Otorhinolaryngology is considered a high-risk specialty, as it is in close contact with upper respiratory tract mucus, secretions, droplets and aerosols during procedures and surgery.
The Corona virus disease 2019 (COVID-19) pandemic has put extraordinary challenges on the medical fraternity. Physicians, surgeons, and other health care workers, performing and aiding in examinations and surgical procedures within the head and neck region, particularly the airway, are at higher risk of exposure and infection from aerosol and droplet contamination.1
Reports note higher rates of infection in otorhinolaryngologists in Wuhan, China as compared to their associates in other specialties. Patel et al. noted that significant numbers of doctors in the world have been infected and/or died, potentially due to exposure to patients with high viral shedding from nasal and oropharyngeal cavities. Otolaryngologists play a crucial role in the management of COVID-19 patients. Due to the innate nature of their work, they are at high risk of exposure, whether working in outpatient clinics, the emergency room or inpatient wards.2 Despite this elevated risk to otorhinolaryngologists, protocols for managing COVID-19-positive patients are enigmatic and have not yet wholly adapted to procedural exposure risk.3
Respiratory infections can be transmitted through droplets of varying sizes. The droplet particles > 5–10 μm in diameter are referred to as respiratory droplets and particles < 5 μm in diameter, as droplet nuclei.4 Airborne transmission differs from droplet transmission as it refers to the presence of microbes within droplet nuclei that remain in the air for long periods of time and are transmitted to others over distances greater than one meter.5
The severe acute respiratory syndrome Corona virus 2 (SARS-CoV-2) is primarily transmitted between people through respiratory droplets and contact routes.5
Such transmission raises critical questions regarding the safety of performing nasal examination and procedures, particularly outpatient endoscopy and endoscopic sinus surgery in patients with possible COVID-19. These concerns arise out of a high degree of uncertainty as to whether endonasal procedures can generate aerosols, and whether COVID-19 can behave as an opportunistic airborne pathogen and transmit infection via these potential aerosols.6,7 These questions have become even more salient as data regarding elevated nasal/nasopharyngeal viral loads of even asymptomatic patients8 and prolonged viral persistence in air has emerged.9 The purpose of this article is to counsel rhinologists on how to modify their conventional practices during the COVID-19 pandemic by prioritising the need of procedures and identifying aerosol generating procedures. The main aim of these advocacies is to protect healthcare workers, particularly rhinologists, against COVID-19 while continuing to provide the best patient care.
MethodsLiterature searchThis manuscript followed the published methodology of developing an evidence-based review with recommendations by Rudmik et al. (2011).10 The ENT Cochrane, Pubmed, DOAJ and Web of Science databases were searched from inception to July 15, 2020 using the terms “COVID-19 and SARS-CoV-2” in conjunction with rhinology, nasal endoscopy, endoscopic sinus surgery and aerosol. Since aerosol and droplet terminology has been used interchangeably in the literature, our search included both terms. Articles mapping to the exploded medical subject heading “nasal endoscopy and SARS-CoV-2” were combined into one group. Medical subject headings “COVID-19 and rhinology” were exploded, and the manuscripts were collected into a second group. Medical subject headings “endoscopic sinus surgery and coronavirus” were combined into a third group. Medical subject headings “aerosol generating medical procedures and coronavirus” were combined into a fourth group. The four groups were then cross referenced.
Inclusion and exclusion criteriaWe evaluated the articles identified by the above search strategy to meet these inclusion criteria: (a) studies that included COVID-19 and (b) rhinology procedures: nasal endoscopy and endoscopic sinus surgery. We have included English articles, clinical or experimental studies involving procedures done in Rhinology. We excluded studies that were opinion papers and articles with abstracts indexed in a searched database but without an associated and accessible full manuscript.
Data extractionExtracted data included study design; epidemiology and population description from where study/observations were reported; mechanisms of infection in the sinonasal cavity; aerosols generating procedures like nasal endoscopy and endoscopic sinus surgery; type of COVID-19 test conducted if applicable; signs and symptoms of COVID-19; COVID-19 risks, safety of healthcare workers and sinonasal anatomy. Review papers were also cross- referenced to ensure all studies were identified.
A total of 14,744 articles were identified using the search criteria. 749 were excluded after reading the text as they were not fulfilling the inclusion criteria. Therefore, in the context of the urgency of the pandemic and the rapid accumulation of new information, 28 articles were finally chosen to prepare this manuscript.
Triage contemplations for rhinologistWho to see for consultation?Before confirming the patient’s appointment, a triage is essential to check the necessity of consultation for an urgent physical examination and to exclude any suspicious signs of COVID-19 in the patient.
The clinical signs of COVID-19 to be considered are:
- 1
Fever (Temp > 38 °C) or feverishness;
- 2
Cough;
- 3
Chest pain caused by coughing;
- 4
Shortness of breath;
- 5
Anosmia ± Ageusia (loss of taste and/or smell) for less than a month;
- 6
Abdominal symptoms (anorexia, diarrhoea, vomiting and abdominal pain);
- 7
High risk of contamination, defined as having been less than a meter away from a COVID-19 positive patient for at least 15 min.
Patients presenting with any of these symptoms should be sent to the suspected COVID-19 emergency department or a specialized center. It is also recommended that the waiting room should be organized to comply with social distancing norms.
Categorisation of procedures on urgency basis during the pandemicGroup A: surgeries that cannot be postponedSinusitis with complications (cavernous sinus thrombophlebitis, meningitis, subperiosteal abscess or orbital abscess), invasive fungal sinusitis, complicated mucocoele (neurological or ophthalmic signs), Very displaced nasal fracture, foreign body in the nasal cavity.
Recommended approach – Emergency surgery (Functional endoscopic sinus surgery, orbital decompression, optic nerve decompression, nasal fracture reduction, endoscopic nasal foreign body removal).11
Group B: risk of prognostic impact if treatment delayed for > 1 monthInverted papillomas, allergic fungal sinusitis in immunosuppressed patients, mucocoele without compression, osteo-meningeal breach (CSF rhinorrhea).
Recommended approach – postpone surgery and reassess the patient after 1 month and adapt the treatment program according to the course of the disease and the spread of the COVID-19 pandemic after consultation with your team. Use of tele-consultation or telephone followup of patients should be prioritized during the pandemic.11
Group C: surgery which can be postponed for a minimum of 6–8 weeks without significant prognostic impactSinonasal polyposis, aspergillus chronic rhinosinusitis, oro-antral fistula, benign lesion to be removed via endonasal route, septorhinoplasty, dacryocystorhinostomy via endonasal route
Recommended approach – Postpone surgery and reassess the patient after 6–8 weeks and adapt the treatment program according to the course of the disease and the spread of the COVID-19 pandemic.11
Decision makingGroup A and COVID-19 positive patient: COVID-19 protocol;
Group A and COVID-19 status unknown: COVID-19 protocol;
Group A and COVID-19 negative patient (interview, PCR and negative scan): Patient operated on in a conventional sector;
Group B or C, irrespective of COVID-19 status: surgery postponed.
Group B should be prioritized over group C once rescheduling is possible, after all patients in group A have been treated.11
Protection at various levels for the rhinologistOtolaryngology procedures should be done according to the triage guidelines, to minimize use of health care resources as well as to minimize risk of spread of infection to Health Care Workers during any procedures.12 However, for emergency procedures and for medical consultancy, the rhinologist should be aware of the risk of infection to themselves and to others, and more importantly, how to attenuate that risk through the proper use of personal protective equipment (PPE). While minimizing the risk of transmission, it is also incumbent on the surgeon to preserve scarce healthcare resources during a pandemic as availability of PPE may be finite.
Personal protective equipment (PPE) levels for health care workers during COVID-19 pandemic13
Level 1 PPE: surgical mask, gown, gloves, face shield or goggles, head cover (optional)
Level 2 PPE: n95/FFP2, water impermeable gown, double gloves, goggles/face shield and head cover, including neck protection
Level 3 PPE: powered air-purifying respirators (PAPRs) or N95/FFP2 + surgical mask, gowns (1. coverall plus gown with integrated hood and boots is preferred 2. water- impermeable gown), double gloves, goggles + (face shield), head cover, including neck protection
For standard patient consultation and routine assessment of patients, during which no interventions are performed, and only medical management is necessary, Level 1 PPE (droplet precautions) is sufficient. Clinical scenarios indicate that Level 2 PPE (airborne precautions) provides sufficient protection to the rhinologist during short duration aerosol generating medical procedures (AGMPs), even if SARS-CoV-2 positive. For long duration AGMPs that are deemed high-risk, in patients known to be SARS-CoV-2 positive, a higher level of protection is recommended, i.e. Level 3 PPE.13
Recommendations during specific outpatient proceduresNasal nebulizer/atomizersVenturi principle atomizers are used to apply topical anesthetics, vasoconstrictors, or other topical medications to the mucous membranes of the nasal cavity. The components of the device are made of plastic and rubber, preventing it from being autoclaved posing a high risk of contamination. With the advent of COVID-19 pandemic, the major concern is the cross contamination through aerosols. This happens when the medication is sprayed into a tapered space, such as the nasal cavity, the jet of the compressed air being very strong tends to rebound. This rebound airflow, combined with the aerosol, easily contacts the tip of the atomizer, potentially contaminating the device and the surrounding.14
One experimental study revealed bacterial contamination of the tip of Venturi atomizers in 9 of the 15 subjects (Table 1).
Elements of included studies on nebulizers/atomizers.
| Author, year | Study design | Subjects (n) | Study group | Study outcomes | Conclusion |
|---|---|---|---|---|---|
| Tseng, 201414 | Experimental study | 15 (30 nares) | Healthy subjects | 1. Bacterial contamination from the nasal vestibule to the atomizer tip through the reverse jet airflow of the atomizer nasal tip. | 1. 60% samples were positive for bacterial growth at the atomizer tip. |
| 2. Dispersion of droplets beyond 10 cm with 0.9 Mpa atomiser and within 10 cm with 0.3 Mpa atomiser. | 2. During the spray process, aerosols were noted travelling backwards through the reversed jet flow and attaching to the nozzle tip, contaminating the tip and the nearby surrounding. |
The atomizer air pressure settings of 0.9Mpa results in a significant number of droplets, carried by reverse jet airflow, to deposit more than 10 cm away in the surroundings, from the struck nasal cavity target. Reducing this pressure to 0.3 Mpa, most of the aerosol depositions were found within the 10 cm range from the nasal cavity target.14
Level of evidence: Grade D – one experimental study.
Directness of evidence: Indirect
Consistency: not applicable (N/A), as one single study could be found.
Precision: imprecise
Recommendations: according to the study, there is production of droplets that are dispersed over a range of 10 cm. Further, it can lead to sneezing and coughing, and therefore can be considered as AGMP. Use of positive displacement atomizers or low-pressure atomizers reduces the reversed jet flow and droplet dispersion. More studies have to be conducted and further research is likely to have an impact on our confidence in the estimate of the effect.
Recommended PPE – level 1
Strength of recommendation: low
Diagnostic nasal endoscopyDiagnostic nasal endoscopy during this pandemic should only be considered for emergency cases and limited to a selective number of benign and malignant cases and severe refractory epistaxis. Even with these cases, we should prefer utilizing imaging modalities (CT, MRI, CT angiography) to diagnose, reserving endoscopy for critical situations where it will have a significant impact on management.
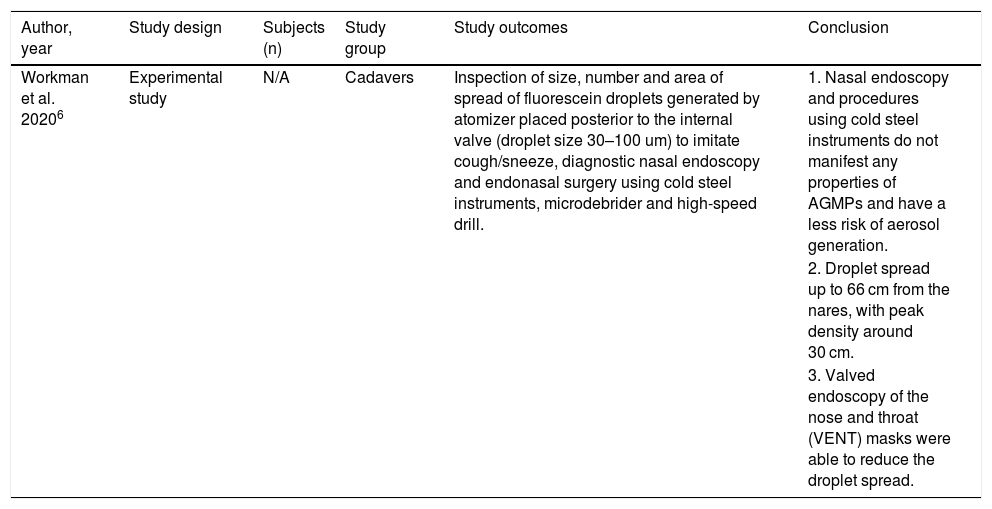
We identified one experimental study evaluating the aerosol generation and their spread risk during nasal endoscopy (Table 2). Workman et al. (2020) simulated potential aerosol generation events using a cadaver with the nasal mucosa coated with fluorescein over an array of possible diagnostic and therapeutic endoscopic procedures.6 The probability of risk of aerosol generation was assessed with the help of a fresh frozen cadaveric model with fluorescein dye solution, a blue-light filter and digital image processor. They concluded that though nasal endoscopy was not an active aerosol generating procedure, the associated coughing and sneezing using an atomization device, producing the aerosols. Activation of the atomizer device, placed through the cribriform plate with the tip positioned posterior to the left internal valve resulted in particle contamination up to 66 cm from the nares (produced droplet size: > 30 um), comprising aerosol contamination. An intact surgical mask and valved endoscopy of the nose and throat (VENT) mask were satisfactory in eliminating all noticeable spread.6
Elements of included studies on nasal endoscopy.
| Author, year | Study design | Subjects (n) | Study group | Study outcomes | Conclusion |
|---|---|---|---|---|---|
| Workman et al. 20206 | Experimental study | N/A | Cadavers | Inspection of size, number and area of spread of fluorescein droplets generated by atomizer placed posterior to the internal valve (droplet size 30–100 um) to imitate cough/sneeze, diagnostic nasal endoscopy and endonasal surgery using cold steel instruments, microdebrider and high-speed drill. | 1. Nasal endoscopy and procedures using cold steel instruments do not manifest any properties of AGMPs and have a less risk of aerosol generation. |
| 2. Droplet spread up to 66 cm from the nares, with peak density around 30 cm. | |||||
| 3. Valved endoscopy of the nose and throat (VENT) masks were able to reduce the droplet spread. |
Level of evidence: grade D – one experimental study.
Directness of evidence: indirect
Consistency: N/A
Precision: precise
Limitations:
- 1
Imitation of irritative sneezing utilized an atomizer, which produced sprays between 30 and 100 μm. Therefore, smaller particles of concern for airborne transmission were not formally assessed.
- 2
Maximum calculated droplet size could have potentially represented the coalescence of several smaller droplets.
Recommendation: according to the study, diagnostic nasal endoscopy should be contemplated as a droplet generating procedure and as a potential AGMP, as it can initiate irritative sneezing and coughing.
Recommended PPE – level 2
Strength of recommendation: moderate
Nasal bleeding and its treatment with nasal packingSevere COVID-19 patients have noticeable hematologic findings consistent with alterations to the coagulation cascade.15 Upper airway instrumentation is commonly required in critically ill patients and often causes mucosal trauma which triggers bleeding.
Anticoagulants for thrombus prevention and extracorporeal membrane oxygenation for acute respiratory syndrome (ARDS) are required in critical COVID-19 patients. They can cause mechanical obstruction of the internal jugular vein or superior vena cava by large cannulas leading to venous congestion, increasing the propensity for persistent bleeding.16
In recurrent or severe bleeding for which medical therapy has failed, the next line of management is nasal packing, cauterization, or both.
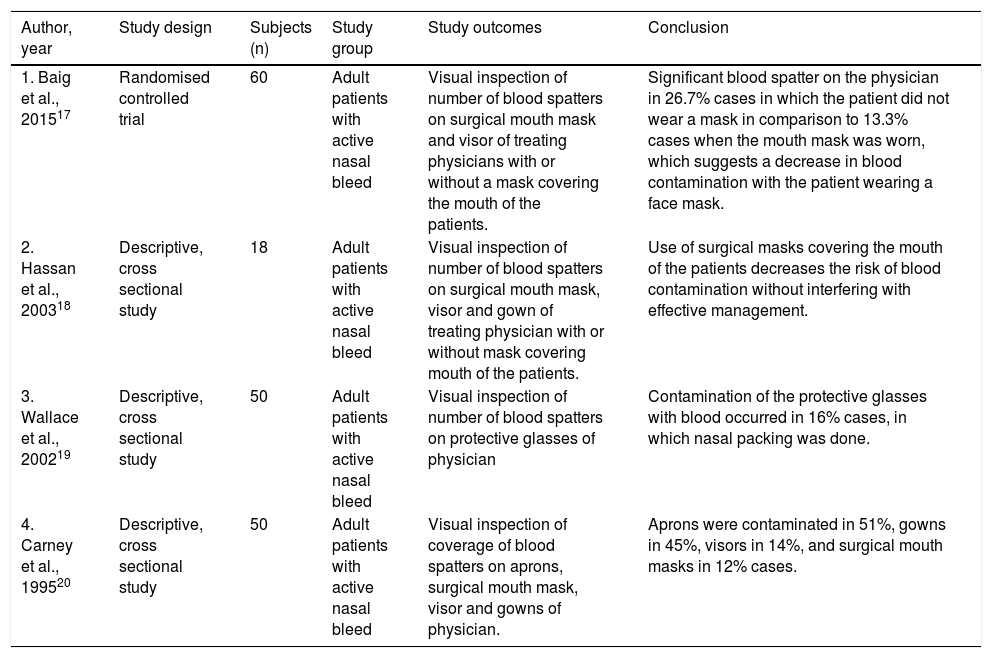
Four studies evaluated the risk of aerosol contamination during the nasal packing by visually examining blood contamination of the personal protective equipment and confirmed the treatment of epistaxis by nasal packing can cause transmission of blood aerosols within close proximity of the patient (Table 3).17–20
Elements of included studies on nasal packing for active nasal bleeding.
| Author, year | Study design | Subjects (n) | Study group | Study outcomes | Conclusion |
|---|---|---|---|---|---|
| 1. Baig et al., 201517 | Randomised controlled trial | 60 | Adult patients with active nasal bleed | Visual inspection of number of blood spatters on surgical mouth mask and visor of treating physicians with or without a mask covering the mouth of the patients. | Significant blood spatter on the physician in 26.7% cases in which the patient did not wear a mask in comparison to 13.3% cases when the mouth mask was worn, which suggests a decrease in blood contamination with the patient wearing a face mask. |
| 2. Hassan et al., 200318 | Descriptive, cross sectional study | 18 | Adult patients with active nasal bleed | Visual inspection of number of blood spatters on surgical mouth mask, visor and gown of treating physician with or without mask covering mouth of the patients. | Use of surgical masks covering the mouth of the patients decreases the risk of blood contamination without interfering with effective management. |
| 3. Wallace et al., 200219 | Descriptive, cross sectional study | 50 | Adult patients with active nasal bleed | Visual inspection of number of blood spatters on protective glasses of physician | Contamination of the protective glasses with blood occurred in 16% cases, in which nasal packing was done. |
| 4. Carney et al., 199520 | Descriptive, cross sectional study | 50 | Adult patients with active nasal bleed | Visual inspection of coverage of blood spatters on aprons, surgical mouth mask, visor and gowns of physician. | Aprons were contaminated in 51%, gowns in 45%, visors in 14%, and surgical mouth masks in 12% cases. |
Level of evidence: grade C – one level 3 study, and three indirect, descriptive cross-sectional studies.
Directness of evidence: indirect
Consistency: all the studies are consistent in the findings.
Precision: imprecise
Limitations: contamination of surroundings during an effective nasal packing depends on the cause of the bleed, expertise of the surgeon and patient compliance.
Recommendation: According to the studies, the surgical treatment of epistaxis inevitably presents risk for droplet emission and viral transmission due to the close contiguity of physician and patient. Nasal packing without anesthesia or sedation is painful; thus, the patient is unable to control involuntary reflexes such as coughing, making it an AGMP.
Moreover, treatment of epistaxis needs prompt medical intervention during this pandemic; as such, the presence of COVID-19 contacts of the patient may not be properly investigated.
Recommended PPE – level 2
Strength of recommendation: moderate
ElectrocauteryCauterization of bleeding point is the next line of treatment for recurrent epistaxis that either requires repacking or is a failure of medical management. Such refractory bleeding is treated using electrocoagulation, particularly in cases with an easily visible anterior bleed.
It coagulates small vessels, controlling bleeding by means of a high-frequency electrical current. Surgical smoke is the gaseous by-product generated by surgical procedures and tools, including electrocautery devices and laser ablation, as a result of the thermal decomposition of tissues. Substantial evidence indicates the presence of numerous chemicals, blood and tissue particles, viruses and bacteria’s in surgical smoke.21–23 Numerous studies prove that the smoke plume is potentially harmful to healthcare workers and patients.21–23
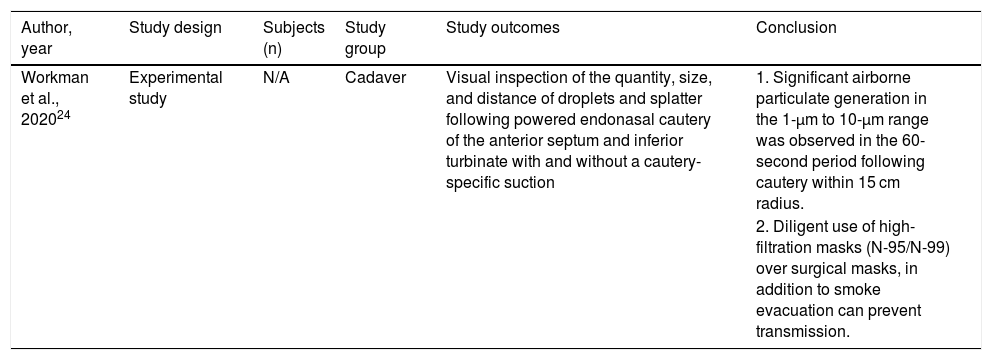
One experimental study done by Workman et al. evaluated the splatter produced by electrocautery in two cadavers (Table 4).24
Elements of included studies on endoscopic electrocautery.
| Author, year | Study design | Subjects (n) | Study group | Study outcomes | Conclusion |
|---|---|---|---|---|---|
| Workman et al., 202024 | Experimental study | N/A | Cadaver | Visual inspection of the quantity, size, and distance of droplets and splatter following powered endonasal cautery of the anterior septum and inferior turbinate with and without a cautery-specific suction | 1. Significant airborne particulate generation in the 1-µm to 10-µm range was observed in the 60-second period following cautery within 15 cm radius. |
| 2. Diligent use of high-filtration masks (N-95/N-99) over surgical masks, in addition to smoke evacuation can prevent transmission. |
Level of evidence: grade D: one experimental study.
Directness of evidence: indirect
Consistency: N/A
Precision: precise
Limitations:
- 1
Experiments were performed in cadaveric specimens and therefore, these results may not be stereotyped for human surgery as increased bleeding and irritative coughing may alter the propensity for airborne particulate production that exceeds the capabilities for evacuation via suction
- 2
Whether SARS-CoV-2 can persist in surgical plumes or is killed by the heat produced by electrocautery is still uncertain.
Recommendation: according to the study, electrocautery should be contemplated as a potential aerosol and infective surgical smoke generating procedure.
Recommended PPE – level 2
Strength of recommendation: low
Endoscopic sinonasal and anterior skull base surgeryThe gold standard for surgery of sinonasal malignancies is to select the approach that is most capable of obtaining negative margins, while minimizing patient morbidity. The endoscopic approach is at least as good as the open approach in obtaining negative margins and carries less morbidity and a shorter hospital stay.25
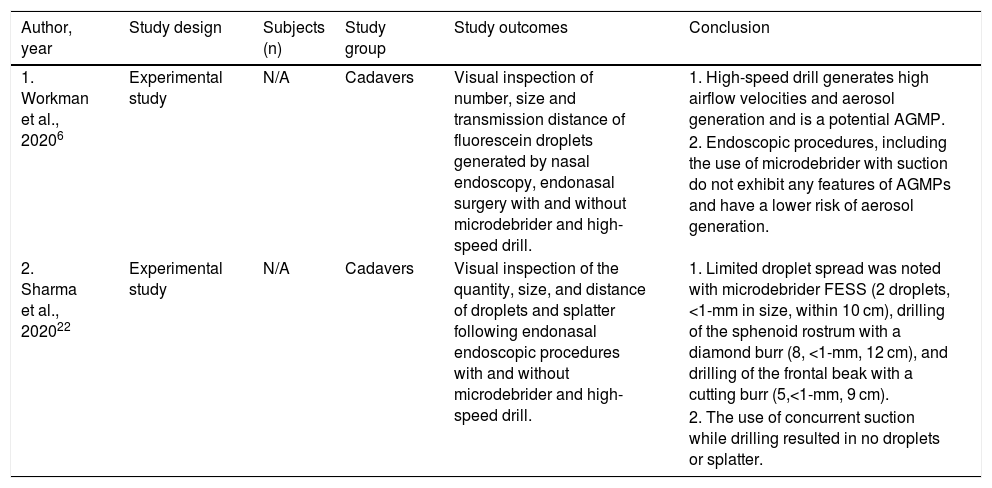
Two experimental studies evaluated the risk of aerosol generation and risk of contamination during various endoscopic endonasal procedures (Table 5).
Elements of included studies on endoscopic sinonasal and anterior skull base procedures.
| Author, year | Study design | Subjects (n) | Study group | Study outcomes | Conclusion |
|---|---|---|---|---|---|
| 1. Workman et al., 20206 | Experimental study | N/A | Cadavers | Visual inspection of number, size and transmission distance of fluorescein droplets generated by nasal endoscopy, endonasal surgery with and without microdebrider and high-speed drill. | 1. High-speed drill generates high airflow velocities and aerosol generation and is a potential AGMP. |
| 2. Endoscopic procedures, including the use of microdebrider with suction do not exhibit any features of AGMPs and have a lower risk of aerosol generation. | |||||
| 2. Sharma et al., 202022 | Experimental study | N/A | Cadavers | Visual inspection of the quantity, size, and distance of droplets and splatter following endonasal endoscopic procedures with and without microdebrider and high-speed drill. | 1. Limited droplet spread was noted with microdebrider FESS (2 droplets, <1-mm in size, within 10 cm), drilling of the sphenoid rostrum with a diamond burr (8, <1-mm, 12 cm), and drilling of the frontal beak with a cutting burr (5,<1-mm, 9 cm). |
| 2. The use of concurrent suction while drilling resulted in no droplets or splatter. |
Workman et al. (2020) analysed the generation of aerosols and the risk of contamination during endoscopic sinonasal procedures. The aerosol generation risk, particle size, and transmission distance are entirely a function of the instrumentation utilized. They observed no droplet generation on performing nonpowered endonasal procedures or with use of the microdebrider. Conversely, use of a high-speed powered drill created high airflow velocities, so it is considered aerosol generating. Contamination was identified with both endonasal and external activation of the drill. The aerosol size produced from drilling was not stated.6
Sharma et al. (2020) in an experimental study analysed the size, number, distance of the droplets and splatter during various therapeutic endoscopic sinonasal procedures. Endonasal procedures including septoplasty, turbinate reduction, and FESS with cold instrumentation were not sources of droplet or splatter contamination. Limited amounts of contamination were visualized following FESS with the microdebrider and powered drilling of the sphenoid rostrum and frontal beak, the observed droplet sizes being less than 1 mm. The use of a concurrent suction while drilling did not result in contamination.26
Level of evidence: grade D – two experimental studies.
Directness of evidence: indirect
Consistency: all the studies are consistent in the findings.
Precision: precise
Limitations:
- 1
The study does not explore the possibility of aerosol formation and contamination for performing procedures for long duration (> 10 min.)
- 2
Only droplets and splatter visible to the human eye were measured.
- 3
Endoscopic visualization and measurement of contamination using a blue light filter was attempted, but unfortunately, it is less sensitive than a glare effect.
- 4
The design model could not perform a complete 360-degree assessment but, only in the cardinal directions surrounding the specimen.
Recommendation: Low aerosol spread with the microdebrider is due to the relatively low speed of rotation and suction in the microdebrider, while high speed drills cause relatively more aerosol generation. The use of high-speed drills appears to be the single greatest risk factor for potential infectious aerosolization during endonasal surgery.6 Aerosol generation can be completely mitigated with the use of concurrent suction in the anterior nasal cavity. However, extreme caution should be used to avoid activating powered instrumentation outside of the nasal cavity, as this was found to cause droplet contamination.26
We concluded that endoscopic surgeries using microdebrider with suction and non-powered instruments (with cautious use of instruments) are non AGMPs, while endoscopic anterior skull base surgeries using drills are AGMPs.
Recommended PPE – level 3
Strength of recommendation: moderate
DiscussionThrough this review, we aim to provide a comprehensive, empirical document for the rhinologist to adopt measures in their practice, based on the limited available data regarding triage, aerosol- generating rhinological procedures and level of personal protection equipment’s, to be followed in the SARS-CoV-2 pandemic.
Multidisciplinary consultation meetings should be encouraged virtually (video or teleconference) to circumvent delays in treating patients and avert participants being exposed to the risk of infection. The PCR tests have a non-negligible false negative rate and are inadequate to define the patient’s COVID-19 status.27 Thus, it is vital that triage is conducted with the patient's personal health interests as the guiding principle.28 We recommend preferring CT-scans over diagnostic nasal endoscopy to monitor pathologies such as nasal polyps or tumors, limiting DNE to grave situations only.
Surgeries like neck exploration for penetrating neck injuries, therapeutic esophagoscopy for corrosive substance ingestion and optic nerve decompression must be performed regardless of the patients’ COVID-19 status. All patients must be assumed to be positive and level 3 PPE usage with appropriate surgical precautions are to be undertaken.
Endonasal procedures using high speed powered rotating instruments such as microdebrider and high-speed drills carry a risk of infectious aerosolization of viral particles such as SARS-CoV-2 and should be considered AGMPs. Nasal endoscopy, nasal packing for epistaxis management and outpatient sinonasal procedures using atomizers tend to induce irritative sneezing and coughing of the patient. This primarily causes droplet transmission, but the resultant formation of clouds of small, inspirable aerosols also poses a risk to the healthcare workers engaged in close vicinity.
The protection of the healthcare worker is important in view of the COVID-19 pandemic. For rhinologists performing AGMPs, guidelines on PPE use are suggested for SARS-CoV-2 positive or suspected patients.1,29 The PPE recommendations are adapted to the nature of the procedure (aerosol generating or non-aerosol generating) and the setting (outpatient service versus operation theater). PPE level 1 is recommended while using atomisers (low pressure or positive displacement) and instilling intranasal steroid sprays. VENT masks should be encouraged for the patients and reuse of the same atomiser discouraged. PPE level 2, including N95 mask is recommended for outpatient endoscopic procedures and nasal packing. Additionally, keeping as much distance as possible, using camera- assisted video endoscopy, and using VENT masks are recommended as the risk of inhaling potentially smaller SARS-CoV-2 infected aerosols should not be neglected. PPE level 3 is recommended in AGMPs viz procedure using high speed drill or microdebrider in COVID-19 positive or suspected patients. An open approach is better in such scenarios, particularly if we can minimize the use of aerosol generating instruments. In case of endonasal endoscopic procedures, use of cold steel instruments should be promoted. Extreme precautions are to be taken while doing endoscopic procedures using powered instruments. Either powered air purifying respirators (PAPR) or body/face/eye protection with N99/FFP3 respirators (99% filtration rate, or 95% if not available) should be used while doing long standing procedures involving risk aerosolization in a suspected or COVID-19 positive patient.27,28
The major limitation of these recommendations is the lack of strong supporting literature and research data. Most recommendations can only be established on the evidence from small, descriptive, experimental studies or indirect retrospective cohort studies. Clinical application of various study results during the current COVID-19 pandemic is still questionable. With the advent of new strategies and knowledge about virus kinetics and with proper precautions there is a need to find out the potential of the coronavirus to survive in aerosols, but also its sensitivity to heat and shear stress during drilling.
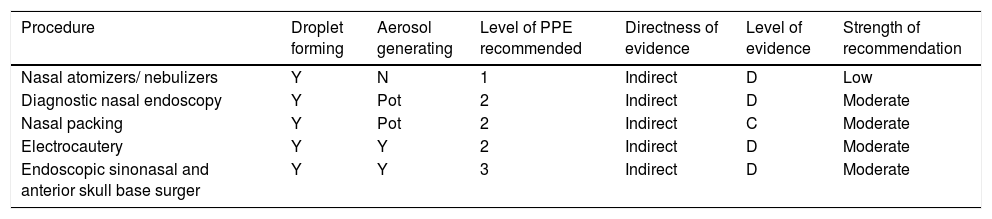
With the available studies, the authors have reached an accord and developed the recommendations to be followed during COVID-19 pandemic for various rhinology procedures; summarized in Table 6.
Summary of rhinology procedures and recommendations.
| Procedure | Droplet forming | Aerosol generating | Level of PPE recommended | Directness of evidence | Level of evidence | Strength of recommendation |
|---|---|---|---|---|---|---|
| Nasal atomizers/ nebulizers | Y | N | 1 | Indirect | D | Low |
| Diagnostic nasal endoscopy | Y | Pot | 2 | Indirect | D | Moderate |
| Nasal packing | Y | Pot | 2 | Indirect | C | Moderate |
| Electrocautery | Y | Y | 2 | Indirect | D | Moderate |
| Endoscopic sinonasal and anterior skull base surger | Y | Y | 3 | Indirect | D | Moderate |
Y, Yes; N, No; Pot, Potential.
Prevailing data suggests that SARS-CoV-2 is primarily transmitted through inhaled aerosols and close contact. Airborne transmission may occur during AGMPs in healthcare settings. HCWs should triage patients on the basis of the need for procedure and then classify the procedure on the basis of whether the procedure is aerosol generating or not. This best practice guidance must of course be adapted in each region according to the evolution of the epidemic and pre-existing arrangements. Level 3 PPE is recommended for HCWs in contact with suspected or confirmed SARS-CoV-2 positive patients, and PAPRs or N95 should be worn by HCWs whenever AGMPs are performed. Selection of the proper level of PPE should be based on a thorough risk assessment of the extent and duration of exposure and the properties of the PPE required for protection. As the world grapples with the devastation of the infectious diseases, the coronavirus pandemic has forced us to rethink our usual standard protocols in the field of head and neck surgery. One fact is clear: much more research is needed to get through this and be well prepared for the future.
Conflicts of interestThe authors declare no conflicts of interest.
Dr Isha Preet Tuli and Dr Sandeep Trehan are responsible for the concept of this paper, wrote the manuscript and made the figure and table outlines. All other authors provided valuable feedback, suggestions, and corrections to improve the quality of the manuscript. All the authors gave final approval of the version to be published and agree to be accountable for all aspects of the work.
Institution: Vardhman Mahavir Medical College and Safdarjung Hospital, New Delhi, India.
Peer Review under the responsibility of Associação Brasileira de Otorrinolaringologia e Cirurgia Cérvico-Facial.