Olfactory changes are quite common in the population, causing a significant impact on the quality of life. Documentation of the olfactory function is essential for the diagnosis, treatment and follow-up of patients with inflammatory diseases of the upper airways, neurodegenerative diseases or viral infections. Among the different existing smell tests, the CCCRC is an inexpensive test, easy to apply, but it has not yet been evaluated on a large scale in the Brazilian population.
ObjectiveTo validate the CCCRC smell test, after adaptation for the Brazilian population, evaluating the performance of healthy volunteers and the stability of the test in retests.
MethodsIn this study, we carried out a cultural adaptation of the CCCRC test to Brazil. To validate and determine the normality scores, we applied the test to 334 healthy volunteers, aged >18 years of age. The retest was also carried out in up to four weeks on 34 additional volunteers to assess validity of the results.
ResultsWhen evaluating the participants’ performance, normosmia and mild hyposmia values were obtained in more than 95% of them. Women (58.4%) showed better accuracy than men (41.6%): p<0.02, and individuals over 60 years of age showed worse performance (median: 6; 75th percentile: 6.5; 25th percentile). The test and retest of the 34 volunteers demonstrated that there was agreement (ICC, intraclass correlation coefficient) considered good in the left nostril (ICC=0.65) and excellent in the right nostril (ICC=0.77) in the combined score.
ConclusionThe CCCRC test adapted to Brazil showed normal values, similar to the originally-described test and validations in other countries, with a high reproducibility rate. Considering the highly favorable cost-benefit ratio, the adapted CCCRC is a very useful tool for measuring olfactory function in the Brazilian population.
Olfaction is one of the five senses that allows humans to interact with the environment through odorous molecules. Based on this sense, we identify situations of danger when we detect molecules that are harmful to health, such as the presence of toxic gases as well as to identify foods that are inappropriate for consumption.1 Moreover, due to the interaction of smell with the limbic system and with upper cortical areas, stimulation of specific odorous receptors promotes diverse emotional responses, which are pleasant or not, creating long-latency olfactory memory.2
Olfactory dysfunctions can negatively impact the individual's physical and psychological health, leading to a worsening in quality of life, anhedonia, mood disorders and decreased appetite, in addition to direct risks, due to the greater propensity for food poisoning and toxic inhalants.3
The main identifiable cause of the loss of olfaction is a viral upper respiratory tract infection (URTI),4 corresponding to about one-third of sudden losses of olfaction, and with a seasonal distribution corresponding to that of viral URTIs.3,5–7 Other common causes are traumatic brain injury, chronic sinonasal diseases, neurological and neurodegenerative diseases.1,4,7,8
As this is a symptom that has high subjectivity among patients, quantitative and qualitative documentation of olfactory function is of utmost importance for patients who present with changes in olfaction. Similarly, it is of fundamental importance to document the olfactory function in patients who will undergo nasal surgeries, such as skull-base surgeries and sinonasal endoscopic surgeries. Additionally, considering the impact of olfaction on some sinonasal diseases, such as allergic rhinitis and chronic rhinosinusitis,7–9 this test would be essential to monitor the response to treatment, whether clinical or surgical.
One of the most widely used smell tests worldwide is the Connecticut Chemosensory Clinical Research Center (CCCRC) test.10 The CCCRC tests both the olfactory threshold and the identification of different odors, allowing for both quantitative and qualitative evaluation of olfaction.10 Additionally, it independently investigates each of the nasal cavities, being able to discriminate the laterality of the olfactory alteration. It is a low-cost test that can be considered for a large-scale clinical application. In spite of this, due to reasons of culturally better known odors or not, it needs to be adapted according to the public to which it will be applied.
In this context, because the CCCRC test has not been widely used for Brazilian individuals, the objectives of this study were to evaluate the scores of healthy Brazilian volunteers at the CCCRC test, after a cultural adaptation to Brazil, and to validate its use for the Brazilian population.
MethodsThis study was approved by the Research Ethics Committee of Hospital das Clínicas da Faculdade de Medicina de Ribeirão Preto under number CAAE: 97227018.5.0000.5440. All volunteers signed an informed consent form. In this study, we applied a non-validated questionnaire on the patients’ personal history (Table 1), followed by the Connecticut smell test adapted to Brazil (adapted CCCRC) in healthy individuals, in two different collection centers (Poupatempo in Ribeirão Preto and Hospital das Clinicas da Faculdade de Medicina de Minas Gerais). As eligibility criteria, we selected individuals over 18 years old, who declared that their olfactory function was present at the time of the evaluation, and the absence of sinonasal complaints compatible with rhinosinusitis. Individuals who declared that their olfactory function was absent, who had a recent upper tract infection, history of severe traumatic brain injury, psychiatric or neurological diseases were excluded.
Clinical questionnaire applied before the smell test.
| No | Yes | |
|---|---|---|
| Do you smoke or have you ever smoked? | ||
| Do you have a good sense of smell? | ||
| Do you have a nasal disease? | ||
| Have you had nasal surgery? | ||
| Do you have a bad smell inside your nose? | ||
| Are you allergic to medications? | ||
| Do you have asthma? | ||
| Do you have allergic rhinitis? | ||
| Have you ever suffered severe trauma (blow) to the head? | ||
| Do you have recurrent nasal bleeding? | ||
| Do you have a disease? Which? |
For the olfactory threshold test, we used butanol (n-butyl alcohol) diluted at seven different concentrations (4%, 1%, 0.4%, 0.1%, 0.05%, 0.01% and 0.005%), as described by Cain.10 The different concentrations of butanol were placed in amber flasks, with the total solution comprising 60mL. The flasks were numbered from 1 to 7, from the highest to the lowest concentration. A flask containing odorless distilled water (flask 8) was used as control.
The threshold test was performed by presenting the individual with two identical 60mL flasks alternately, one containing distilled water and the other a butanol solution. With their eyes closed, the subjects were instructed to occlude one of the nostrils while the flask was placed in the patent nostril, asking the subject to inhale gently to identify the presence of the odor. If the individual could not identify the odor, flasks with more concentrated solutions of butanol were presented consecutively, while maintaining the alternation with the flask of distilled water. The testing was started by exposing the individual to the lowest concentration of butanol (0.005%) until the detection of the flask with the subsequent higher concentration, with retesting being carried out when necessary. Two correct identifications of the flask containing the odorant determined the minimum concentration defined as the individual's olfactory threshold for the tested nasal cavity. The same steps were repeated in the contralateral nasal cavity. The score ranged from 0 to 7 points, with 0 being the individual who was unable to identify the flask containing the odorant at any concentration, and 7 being the individual who identified the odorant in the flask with the lowest concentration.
A total of 8 substances were used for the substance identification test: powdered coffee, powdered cinnamon, talcum powder (Johnson&Johnson® baby powder), paçoca, a typical Brazilian candy made of ground peanuts and sugar (Paçoquita®), chocolate powder (Nescau®), neutral soap (Palmolive®) and mothballs. To adapt the test to the Brazilian reality, we replaced the peanut butter in the original test for paçoca, as it is a more nationally recognized product. The substances were placed in sealed jars, with a lid and a black label, containing equivalent proportions (10g/flask).
For the identification of odors, the individuals previously received a list that contained the name of the eight substances present in the test, in addition to the name of eight distracting substances (Table 2). The subjects, with their eyes closed, were instructed to occlude one nostril while the flask was positioned in the other patent nostril. Before opening each flask, the flask was gently agitated to homogenize the odorous substances inside. For each flask presented to the individuals, they had to name one of the substances on the list. In case of doubt, each flask could be presented again, as well as the list itself. After testing the 8 substances in one nostril, the same steps were repeated for the contralateral nostril, presenting the flasks at random to prevent the individuals from memorizing the order. The trigeminal nerve function was tested at the end of the test with the presentation of menthol (Vicks® VapoRub), but the identification of this substance was not included in the final score. The identification test score was obtained by scoring from 0 to 7 for each nasal cavity, according to the number of odors that were correctly identified.
The final test score was calculated using the arithmetic mean of the correct answers in the identification of the substances (scores from 0 to 7) with the score obtained from the identification of the butanol threshold for each nasal cavity. The mean was defined for each side, with the olfactory threshold classified as normosmia (score 6–7), mild hyposmia (5–5.75), moderate hyposmia (4–4.75), severe hyposmia (2–3.75) and anosmia (0–1,75) (Table 3).
Olfactory performance classification – CCCRC test.10
| Normosmia | 6–7 |
| Mild hyposmia | 5–5.75 |
| Moderate hyposmia | 4–4.75 |
| Severe hyposmia | 2–3.75 |
| Anosmia | 0–1.75 |
After the test application phase to determine the scores in individuals without complaints, a new sample of 34 participants was added to verify the degree of individual agreement in the test–retest. This group, corresponding to 10.1% of the total sample, consisted of individuals with the same eligibility criteria and who had not been previously tested. We applied the test twice in this sample, with an interval of one to four weeks between them, to assess the reproducibility of the scores. Between applications, the thresholds reached, and the substances contained in each flask were not disclosed. We followed the same protocol for the threshold assessment and identification of the previously mentioned substances.
The data were analyzed using the BM SPSS Statistics v. 20. Categorical variables were expressed as absolute (n) and relative (%) frequency. For the numerical variables, the median and percentiles, and minimum and maximum values were calculated. To compare the results of the smell test, the Mann–Whitney non-parametric test for independent samples was applied. The test was chosen based on the normality tests of Shapiro–Wilk and Kolmogorov–Smirnov, which ruled out a normal distribution in the assessed groups. We considered it to be statistically significant when p-value was <5%. To evaluate the test–retest, the degree of agreement of the combined scores was determined, using the statistical analysis of the intraclass correlation coefficient.
ResultsThe Connecticut Olfactory Test (CCCRC) adapted to Brazil was applied to 334 volunteers. The participants’ age ranged from 18 to 76 years, with a mean age of 39.9 years (±14.5).
The result of the medians of the combined scores in each nostril was within the normosmia threshold (results >6), as shown in Table 4. There was no difference between the results of each nostril. The evaluation of the integrity of the trigeminal nerve receptors was positive in 98.5% in the right nasal cavity and 97.3% in the left one.
Results of the CCRC teste adapted to Brazil.
| Evaluation | Median | Minimum | 25th percentile | 75th percentile | Maximum |
|---|---|---|---|---|---|
| Right side | |||||
| Olfactory threshold | 7.0 | 2.0 | 6.0 | 7.0 | 7.0 |
| Smell identification | 7.0 | 0.0 | 6.0 | 7.0 | 7.0 |
| Combined score | 6.5 | 2.5 | 6.0 | 7.0 | 7.0 |
| Left side | |||||
| Olfactory threshold | 7.0 | 2.0 | 6.0 | 7.0 | 7.0 |
| Smell identification | 7.0 | 0.0 | 6.0 | 7.0 | 7.0 |
| Combined score | 6.5 | 2.5 | 6.0 | 7.0 | 7.0 |
The final classification of the volunteers resulted in normosmia in 82.6% of the right nostrils and 83.55% of the left nostrils (Table 5). If this value is added to the tested ones that were classified as mild hyposmia, we reach 95.8% and 96.7% in the right and left nostrils, respectively.
Regarding the clinical variables obtained in the questionnaire (Table 6), no association was found regarding the olfactory performance in patients who reported smoking, allergic rhinitis, previous nasal surgery, traumatic brain injury, asthma, drug allergy, and epistaxis. The only significant result was related to the performance of patients who reported metabolic comorbidities. It is worth mentioning that the majority of the positive responses in the questionnaire were from fewer than 20 participants.
Assessment of the association between different clinical variables and test performance.
| Variable | Right side | Left side | ||||||
|---|---|---|---|---|---|---|---|---|
| Median | 25th percentile | 75th percentile | p-value | Median | 25th percentile | 75th percentile | p-value | |
| Smoking | ||||||||
| Yes (n=66) | 6.50 | 6.00 | 7.00 | 0.26 | 6.50 | 6.00 | 7.00 | 0.36 |
| No (n=268) | 6.50 | 6.00 | 7.00 | 6.50 | 6.00 | 7.00 | ||
| Preserved olfaction | ||||||||
| Yes (n=322) | 6.50 | 6.00 | 7.00 | 0.93 | 6.50 | 6.00 | 7.00 | 0.40 |
| No (n=12) | 6.75 | 5.50 | 7.00 | 6.25 | 5.37 | 7.00 | ||
| Nasal disease | ||||||||
| Yes (n=12) | 6.75 | 6.00 | 7.00 | 0.57 | 6.50 | 5.75 | 7.00 | 0.78 |
| No (n=322) | 6.50 | 6.00 | 7.00 | 6.50 | 6.00 | 7.00 | ||
| Nasal surgery | ||||||||
| Yes (n=10) | 6.25 | 6.00 | 7.00 | 0.70 | 6.00 | 6.00 | 6.00 | 0.92 |
| No (n=324) | 6.50 | 6.00 | 7.00 | 6.50 | 6.00 | 7.00 | ||
| Drug allergies | ||||||||
| Yes (n=28) | 6.50 | 6.50 | 7.00 | 0.31 | 6.50 | 6.50 | 7.00 | 0.10 |
| No (n=324) | 6.50 | 6.00 | 7.00 | 6.50 | 6.00 | 7.00 | ||
| Bad smell | ||||||||
| Yes (n=9) | 6.50 | 6.00 | 7.00 | 0.77 | 6.00 | 5.50 | 6.50 | 0.07 |
| No (n=325) | 6.50 | 6.00 | 7.00 | 6.50 | 6.00 | 7.00 | ||
| Asthma | ||||||||
| Yes (n=10) | 6.50 | 6.00 | 7.00 | 0.72 | 6.50 | 6.00 | 7.00 | 0.81 |
| No (n=324) | 6.50 | 6.00 | 7.00 | 6.500 | 6.00 | 7.00 | ||
| Rhinitis | ||||||||
| Yes (n=66) | 6.50 | 6.00 | 7.00 | 0.19 | 6.50 | 6.00 | 7.00 | 0.29 |
| No (n=268) | 6.50 | 6.00 | 7.00 | 6.50 | 6.00 | 7.00 | ||
| Head trauma | ||||||||
| Yes (n=20) | 6.50 | 6.50 | 6.75 | 0.97 | 6.50 | 6.00 | 6.75 | 0.50 |
| No (n=314) | 6.50 | 6.00 | 7.00 | 6.50 | 6.00 | 7.00 | ||
| Nasal bleeding | ||||||||
| Yes (n=9) | 6.50 | 6.00 | 7.00 | 0.92 | 6.50 | 6.00 | 7.00 | 0.92 |
| No (n=325) | 6.50 | 6.00 | 7.00 | 6.50 | 6.00 | 7.00 | ||
| Nasal disease | ||||||||
| Yes (n=49) | 5.50 | 6.50 | 6.50 | <0.01 | 5.50 | 6.50 | 7.00 | 0.01 |
| No (n=285) | 6.50 | 6.00 | 7.00 | 6.00 | 6.50 | 7.00 | ||
Value obtained by calculating the Mann–Whitney non-parametric test (independent samples).
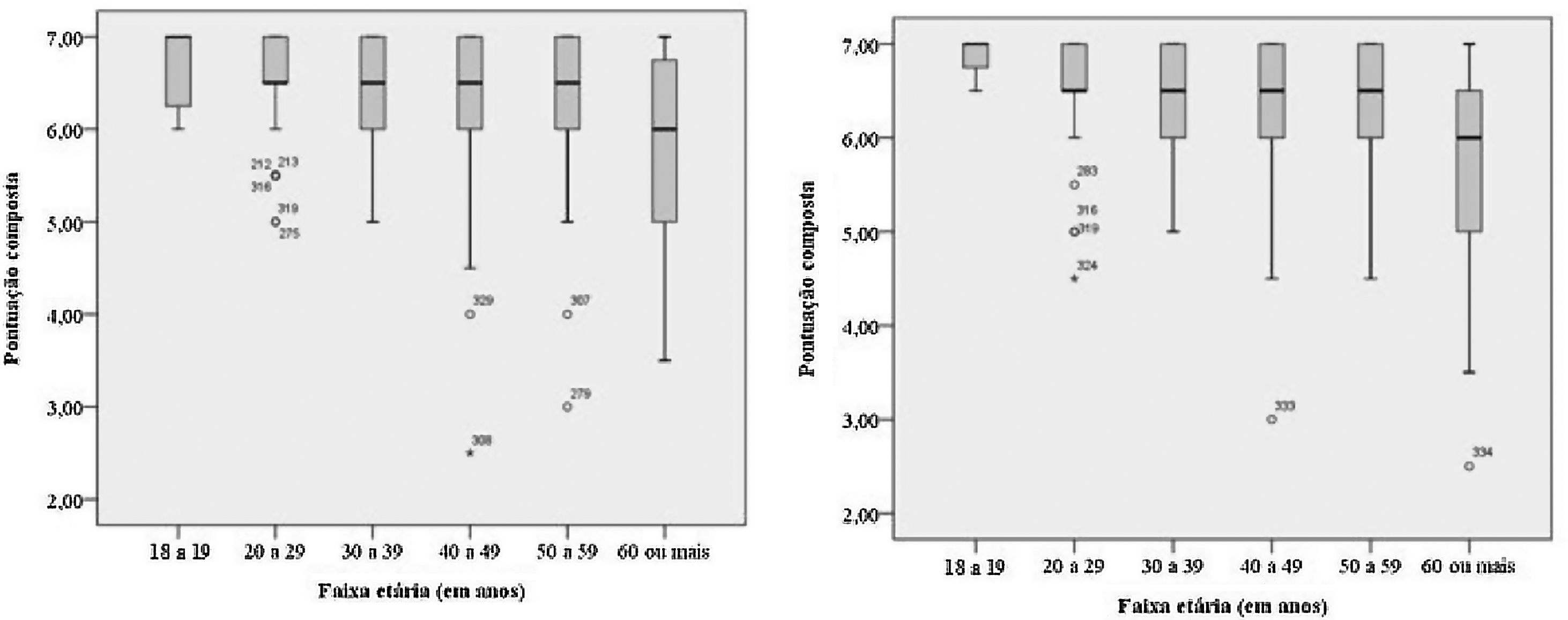
The age distribution verified in our study was compatible with the national age distribution, as well as with the distribution between men and women (ratio of 0.71/1). The mean family income in our sample also showed a correspondence when compared to the income of the Brazilian population according to Brazilian Institute of Geography and Statistics (IBGE, Instituto Brasileiro de Geografia e Estatística) (Table 7). Olfactory performance was lower in individuals over 60 years old (median: 6; 75th percentile: 6.5; 25th percentile: 5) compared to other age groups (median of age ranges 20–30, 30–40, 40–50, 50–60 years: 6.5; median of age range 18–20 years: 7), with a decrease in the scores (Fig. 1).
Sociodemographic variables of the Brazilian volunteers submitted to the CCCRC test adapted to Brazil.
| Variable | n | % |
|---|---|---|
| Gender | ||
| Female | 195 | 58.4 |
| Male | 139 | 41.6 |
| Ethnicity | ||
| White | 178 | 53.3 |
| Mixed-race | 105 | 31.5 |
| Black | 49 | 14.7 |
| Yellow | 1 | 0.3 |
| Age range | ||
| 10–19 years | 11 | 3.3 |
| 20–29 years | 89 | 26.7 |
| 30–39 years | 77 | 23.1 |
| 40–49 years | 62 | 18.6 |
| 50–59 years | 55 | 16.5 |
| 60 or older | 39 | 11.7 |
| Level of schooling | ||
| Incomplete Elementary School | 48 | 14.4 |
| Complete Elementary School | 18 | 5.4 |
| Incomplete High School | 15 | 4.5 |
| Complete High School | 122 | 36.5 |
| Incomplete College/University | 47 | 14.1 |
| Complete College/University | 84 | 25.1 |
| Family incomea | ||
| Up to 2 minimum wages | 151 | 45.2 |
| From 2 to 4 minimum wages | 91 | 27.2 |
| From 4 to 10 minimum wages | 39 | 11.7 |
| From 10 to 20 minimum wages | 13 | 3.9 |
| Above 20 minimum wages | 1 | 0.3 |
| Undeclared income or individual without income (dependent) | 39 | 11.7 |
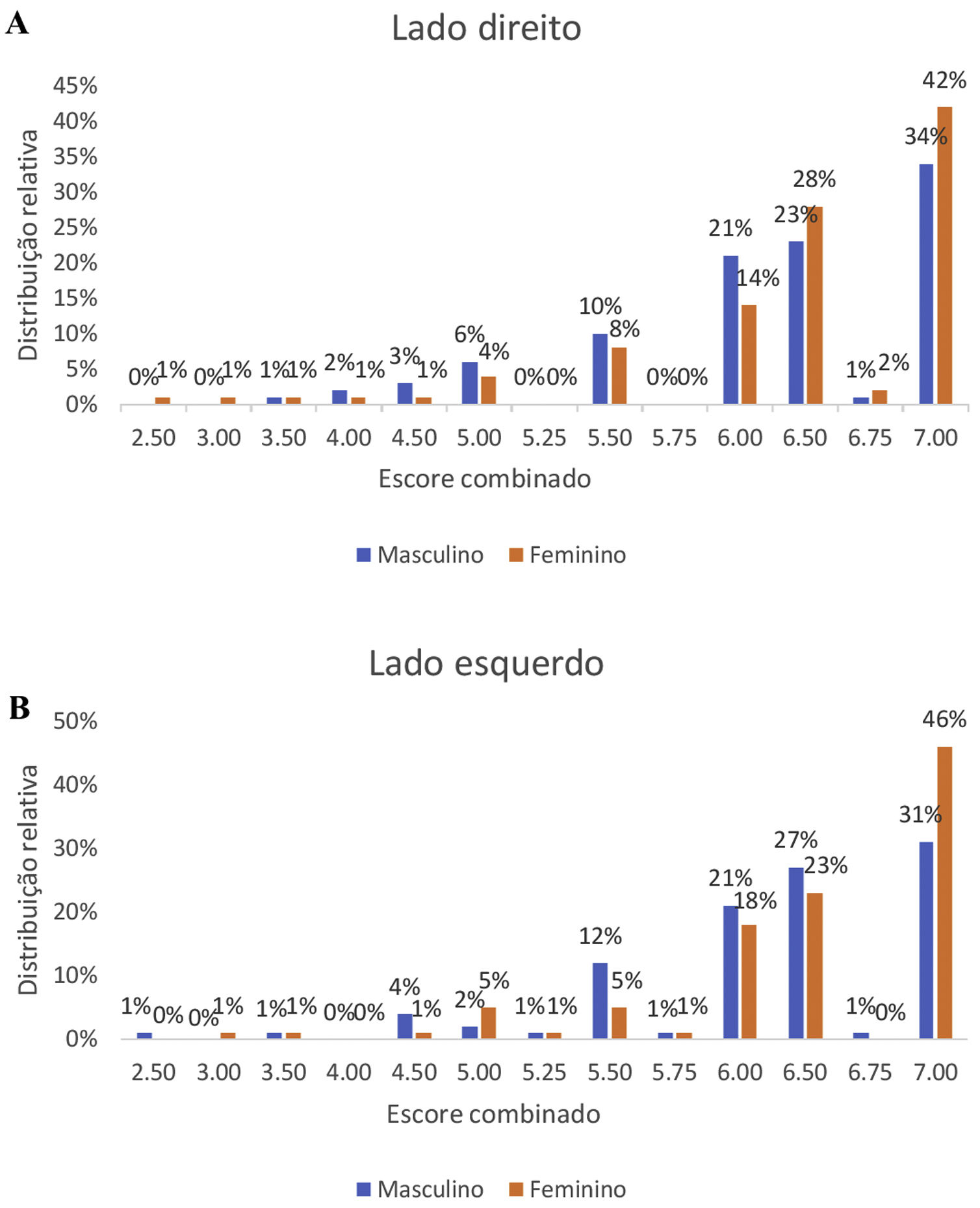
When comparing the olfactory performance in relation to gender, we observed a better performance of female volunteers (median: 6.5) compared to males (median: 6.5), with p-value <0.02 (Fig. 2).
The intraclass correlation of the test–retest in the combined score was 0.77 (95% CI, 0.54–0.88) on the right and 0.65 (95% CI, 0.29–0.82) on the left, considered as excellent and good, respectively. The mean number of days after the test when the retest was applied was 9.9 days (Table 8).
Analysis of the CCCRC test–retest agreement between the study participants (n=34).
| Variable | Test median | Retest median | ICC | 95% CI ICC |
|---|---|---|---|---|
| Olfactory threshold | ||||
| Right | 7.00 | 7.00 | 0.76 | 0.53–0.88 |
| Left | 6.50 | 7.00 | 0.69 | 0.38–0.85 |
| Smell identification | ||||
| Right | 7.00 | 7.00 | 0.91 | 0.81–0.95 |
| Left | 7.00 | 7.00 | 0.66 | 0.33–0.83 |
| Combined score | ||||
| Right | 6.50 | 6.50 | 0.77 | 0.54–0.88 |
| Left | 6.50 | 6.50 | 0.65 | 0.29–0.82 |
ICC, intraclass correlation coefficient; 95% CI, 95% confidence interval.
Interpretation of the intraclass correlation coefficient: correlation: >0.75 – excellent; 0.6–0.75 – good; 0.4–0.6 – regular; <0.4 – weak.
The nose, as an anatomical structure of the upper respiratory tract, has olfactory chemoreception and signaling and signal transduction as one of its most intricate mechanisms. The anatomy involving the lateral wall, with the division into nasal turbinates, lower, middle, upper and possibly supreme parts, nasal septum and external cartilaginous pyramid, has characteristics that make the nasal function perfectly when balancing the filtering, humidifying and air-heating functions. The olfactory epithelium consists of olfactory receptor neurons (ORN), basal cells, support cells and microvillar cells. Therefore, anatomical, mechanical or sensorineural alterations may be related to olfactory changes.
Currently, there are some validated smell tests in Brazil. One of them, the smell identification test previously called UPSIT11,12 is easy to use, self-applied, and consists of the identification of 40 different substances arranged in cards in which, after scraping a tape, eliminate micro-encapsulated odorous particles, requiring a forced response out of four options. As disadvantages, the test consists only of the identification of odors, not the threshold, and it has a high cost, which makes it difficult to use it on a large scale and in public health services. Another test is the Sniffin’ Sticks,13 which assesses both the threshold and the identification of odors and has been validated even for the pediatric population14; however, this test is also expensive, which prevents its routine use in the Brazilian public health service.
In search for a more viable test, one that is both low-cost and easy to apply, we chose to validate the CCCRC in Brazil. The CCCRC has some advantages to identify both the olfactory threshold as well as quantifying the identification of seven specific odors. Moreover, it allows the identification of each one of these characteristics in each nostril, separately. Another advantage of the CCCRC is its low cost; the test can even be reused in other people within a period of up to 90 days. However, one of the disadvantages in relation to the UPSIT test, is that the CCCRC cannot be self-applied, requiring a trained professional for the procedure, which takes a little longer for it to be applied (around 15min).
Through the random selection of individuals at the research sites, we attained an adequate representativeness of economic class and age distribution,15 and even with a considerable percentage representation of the elderly population, there were few results of olfactory alterations worse than mild hyposmia.
We previously considered that social differences could result in differences in the test performance, since economically privileged strata could have greater opportunities to be in contact with a greater diversity of odors compared to the less favored social strata. The relevance of the social sampling is related to the fact that a nationally adequate olfactory test must be considered for its accuracy in relation to the chosen substances. Therefore, the diversity of the studied sample and its representativeness in relation to the Brazilian society, combined with the volunteers’ performance, demonstrate that, in people without olfactory complaints, there is a high percentage of correct identification of the tested substances and butanol detection, equivalent to international data.
The value of the CCCRC Brasil test score obtained in the sample is compatible with the results of validations of the CCCRC in other populations16,17 and similar to those demonstrated in the original study of the CCCRC test.10 Thus, we were able to demonstrate that the exchange of peanut butter for paçoca showed an excellent performance for the Brazilian adaptation of the test, since peanut butter is not well known among Brazilians, especially those of less privileged social classes. Although individuals who had olfactory complaints were excluded from the study, the performance analysis, when related to age, showed a decrease in the score of those over 60 years old, corroborating the literature that says that elderly individuals may have worse olfactory discrimination power, despite the absence of complaints.17 Such fact can be related to the low perception of the limitation, or perhaps the little stimulation that the elderly population has in their routine.
As for the test performance related to gender, we observed that women have better olfactory performance than men, similarly to other previous studies that raised the possibility of the association with hormonal protection and less exposure to environmental factors.18
No significance was observed regarding the correlation of test performance with the applied questionnaire, except for comorbidities. Volunteers with metabolic comorbidities had a lower score (p<0.05). However, the objective of the study was not to evaluate this particular population, leading to a small number of positive responses, which may have influenced this specific result.
Regarding the intraclass correlation coefficient, the variables of olfactory threshold, odor identification and combined scores, all on the right side, showed an excellent correlation, whereas those on the left side were considered good, making the test reliable and reproducible when applied around 10 days later.
ConclusionThe CCCRC test adapted to Brazil showed stability in relation to the retesting. The exchange of peanut butter for paçoca did not interfere with the expected final result, in comparison with other validations and the original study. Considering the Brazilian reality, the CCCRC smell test adapted to Brazil becomes a useful tool for olfactory testing in our population.
Conflicts of interestThe authors declare no conflicts of interest.
Peer Review under the responsibility of Associação Brasileira de Otorrinolaringologia e Cirurgia Cérvico-Facial.