Alterations in the vocal folds that involve volume reduction and glottal closure failure result in exaggerated air escape during speech. For such situations, the use of implants or grafts of different materials has been proposed.
ObjectiveTo define the effect of sugarcane biopolymer gel when implanted in the vocal folds of rabbits.
MethodsThis was an experimental study. The vocal folds of rabbits injected with sugarcane biopolymer and saline solution were histologically evaluated after 21 and 90 days.
ResultsMild to moderate inflammation and increased volume were observed in all vocal folds injected with biopolymer, when compared to controls. There were no cases of necrosis or calcification.
DiscussionThis study showed higher inflammatory reaction in cases than in controls and biopolymer biointegration to the vocal fold. This fibrogenic response with absence of epithelial repercussions suggests that the biopolymer in its gel form can be bioactive and preserve the normal vibratory function of the epithelium.
ConclusionWe show that in spite of producing an inflammatory reaction in vocal fold tissues, the material remained in vocal fold throughout the study period.
As alterações das pregas vocais que envolvem redução do seu volume e falha no fechamento glótico causam um escape exagerado de ar durante a fonação. Para essas situações, tem sido proposta a utilização de implantes ou enxertos de materiais diversos.
ObjetivoDefinir o comportamento do biopolímero de cana-de-açúcar implantado nas pregas vocais de coelhos.
MétodosTrata-se de um estudo experimental. Avaliaram-se histologicamente as pregas vocais de coelhos injetadas com biopolímero de cana-de-açúcar e solução fisiológica após 21 e 90 dias.
ResultadosFoi observada a presença do biopolímero, reação inflamatória leve a moderada e aumento de volume em todas as pregas vocais injetadas em relação às de controle. Não houve casos de necrose ou calcificação.
DiscussãoEste trabalho mostrou maior reação inflamatória nos casos que os controles além de biointegração do material na prega vocal. Essa resposta fibrogênica com ausência de repercussões epiteliais pode nos sugerir que o biopolímero em sua forma de gel pode ser bioativo e preservar as funções vibratórias normais do epitélio.
ConclusãoNeste trabalho, mostramos que apesar de produzir uma reação inflamatória nos tecidos das pregas vocais o material perdurou na prega vocal durante todo o período de estudo.
When affected by congenital or acquired anomalies the vocal folds can upset glottal closure during phonation; in these circumstances the persistence of a glottal gap of variable size and shape of results in an exaggerated leak of exhaled air, which alters or prevents vocal fold vibration and alters the voice.1–4 In these situations, the use of implants or grafts of different materials have been proposed to increase the affected vocal fold volume and thus improve glottal coaptation during phonation.5
The choice and use of the ideal material depends on several factors, such whether it is easy to obtain, quantity available, low donor morbidity, immunogenic and mutagenic characteristics, susceptibility to infection, malleability, absorption rate and cost.6
Currently there is a growing interest in the production through fermentation processes, as well as the scientific and clinical use of polysaccharides obtained byusing microorganisms capable of converting certain substances into gels, viscous solutions or membranes (biopolymers).7
An exopolysaccharide biopolymer obtained by fermentation of sugarcane molasses by a bacterium (Zoogloea SP) was synthesized after 1990.8 Its low cytotoxicity, biocompatibility and low production cost stimulated research on this material.9 Its use has been widely studied in several research projects in the reconstruction of different tissues of the human body.9–15
No study had yet been performed to evaluate the possibility of using sugarcane biopolymer (SCB) as graft material in the vocal folds.
ObjectivesThis study aimed to analyze tissue reaction 21 and 90 days after SCB injection in the vocal folds of rabbits, describing the presence of the material and the inflammatory process, as well as vocal cord thickness.
MethodsThis is a prospective randomized case-control experimental study.
A total of 12 adult male California rabbits, ranging in weight from 1.80kg to 3.7kg, with good nutritional status and median weight of 2.45kg were selected. The rabbit was chosen because the vocal folds of rabbits are similar to human vocal folds. The vivisection of the animals complied with the requirements of Federal Law No. 9605 art. 32 and Decree No. 3179 art. 17 dated from 09/21/1999. The Ethics Committee on Animal Experimentation of the institution recommended full approval, under number 23076.003670/2009-46.
Surgical procedures were performed under general anesthesia without tracheal intubation with laryngeal exposure of rabbits by oral route under direct view. One vocal fold received an injection of 0.1mL of SCB in gel form (1% polysaccharide hydrogel). The contralateral vocal fold was submitted to the same procedure with 0.1mL of saline solution. There was no technical difficulty in performing the procedures in rabbits.
For the histological analysis, the larynx was excised and divided into two hemi-larynges. The animals were divided into two temporal groups, each with five rabbits, randomly selected. Group definition was carried out by drawing lots after the surgical procedure. The first group was euthanized 21 days after the procedure (group I) and the second after 90 days (group II).
The rabbits were euthanized with an overdose of the anesthetic drugs xylazine and ketamine.
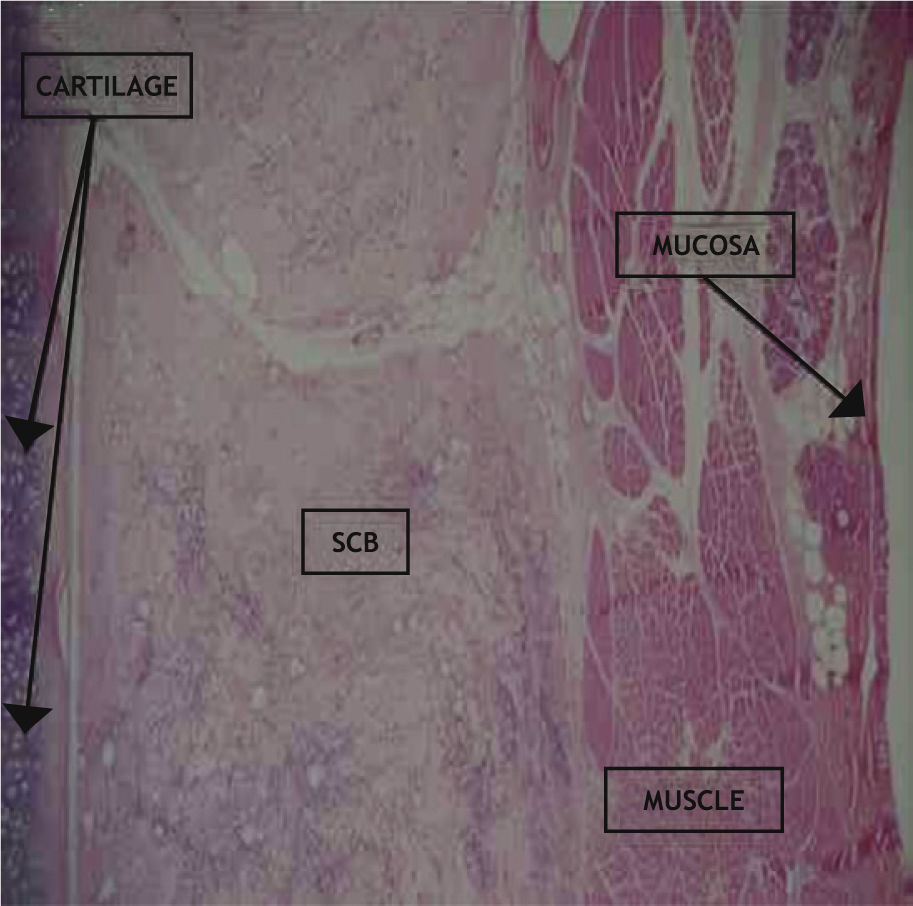
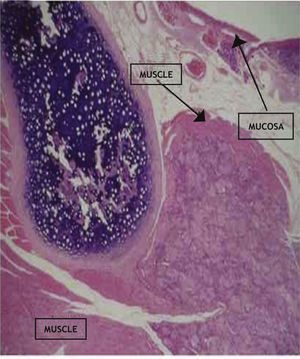
The choice of the vocal fold to be submitted to SCB implantation was randomly assigned before the surgery. The biopolymer was inserted between the vocal muscle and laryngeal cartilage (Fig. 1). The vocal fold submitted to saline solution injection only was considered the control vocal fold. Two animals died; the first in the immediate postoperative period and the second during surgery, probably due to deep anesthesia. Neither of these animals was used in the study.
The two hemi-larynges were evaluated by a single examiner, blinded to the group to which they belonged. The surgical specimens were fixed in 10% formalin for 24hours and three-micrometers thick coronal sections were performed, going through the membranous portion of the vocal fold. The sections were stained with hematoxylin-eosin (HE).
The inflammatory process intensity in the vocal fold submitted to biopolymer injection was compared to the contralateral vocal fold in the different study periods: 21 days (group I) and 90 days (group II) after the procedure.
A comparative study was performed for the presence of fibrosis, lymphocytic inflammatory infiltrate, polymorphonuclear infiltrate, giant cell predominance, cell types (neutrophils, mast cells, lymphocytes, plasma cells, eosinophils, and histiocytes), presence of biopolymers, calcification, necrosis and angiogenesis and thickness measurements in its greatest width in the vocal fold with SCB in relation to the control fold in both study periods.
The histological variables used for lymphomononuclear inflammatory infiltrate were absent (0), grade I (<10% of the area occupied by inflammatory cells), grade II (11%-50%), grade III (>50%). For polymorphonuclear inflammatory infiltrate: absent (0), grade I (<10% of the area occupied by inflammatory cells), grade II (11%-50%) and grade III (>50%). For giant cells, the mean number of giant cells per high-power field was used (magnification×40).
Qualitative variables for fibrosis were: score 0 (absent), score 1 (mild, rare fibers in up to 10% of the area) score 2 (moderate, dispersed fibers, young, not modeled, between 10% and 75%) and score 3 (intense, mature fibers, modeled, 75% to 100%). Biopolymer persistence was present or absent; calcification (present or absent), necrosis (present or absent); angiogenesis was evaluated for the mean number of vascular spaces per high-power field (magnification×40).
Vocal fold thickness was measured from the point of greatest width of the vocal fold from the cartilage to the epithelial surface, using a ruler overlapping the lamina.
To compare the qualitative variables (intensity of the infiltrate, cell predominance, fibrosis) between the groups, Fisher exact test was used, which tests the homogeneity of the two groups in relation to data distribution of responses at the different valuated levels. To compare the quantitative variables (angiogenesis, giant cells and vocal fold thickness), the Mann-Whitney U-Wilcoxon test was used, which is a nonparametric test for the evaluation of two independent populations when one cannot classify the sample as having normal distribution.
ResultsTables 1 and 2 show the findings of Group I (21 days). Tables 3 and 4 show the findings in group II (90 days).
Histological characteristics found in Group I - 21 days – Vocal fold with biopolymer.
| Rabbit | Thickness | Polymer | LMNII | PMNII | GC | Fibrosis | Angiogenesis |
|---|---|---|---|---|---|---|---|
| 1 | 2.2 | + | 1 | 0 | 13.4 | 1 | 1,5 |
| 2 | 3.1 | + | 2 | Eos 1 | 1.7 | 0 | 3,9 |
| 4 | 4.0 | + | 2 | Eos 3 | 2.6 | 1 | 3,7 |
| 5 | 4.0 | + | 1 | 0 | 13.6 | 1 | 3,4 |
| 10 | 2.2 | + | 1 | 0 | 64.0 | 0 | 2,0 |
LMNII, lymphomononuclear inflammatory infiltrate (0-3); PMNII, polymorphonuclear inflammatory infiltrate (0-3), Eos, cellular predominance of eosinophils, GC (giant cells), mean number of giant cells per high-power field (40 ×); Fibrosis, intensity of fibrosis observed; Polymer, presence or absence of the biopolymer; Angiogenesis, mean number of vascular spaces per high-power field (40 ×); thickness, measurement of the vocal fold thickness at its point of greatest width.
Histological characteristics found in Group I - 21 days – Control vocal fold.
| Rabbit | Thickness | Polymer | LMNII | PMNII | GC | Fibrosis | Angiogenesis |
|---|---|---|---|---|---|---|---|
| 1 | 1.8 | 0 | 0 | 0 | 0 | 0 | 0 |
| 2 | 1.1 | 0 | 0 | 0 | 0 | 0 | 0 |
| 4 | 2.4 | 0 | 0 | 0 | 0 | 0 | 0 |
| 5 | 2.4 | 0 | 0 | 0 | 0 | 0 | 0 |
| 10 | 2.4 | 0 | 0 | 0 | 0 | 0 | 0 |
LMNII, lymphomononuclear inflammatory infiltrate (0-3); PMNII, polymorphonuclear inflammatory infiltrate (0-3), Eos, cellular predominance of eosinophils, GC (giant cells), mean number of giant cells per high-power field (40×); Fibrosis, intensity of fibrosis observed; Polymer, presence or absence of the biopolymer; Angiogenesis, mean number of vascular spaces per high-power field (40 ×); thickness, measurement of the vocal fold thickness at its point of greatest width.
Histological characteristics found in Group II - 90 days - Vocal fold with biopolymer.
| Rabbit | Thickness | Polymer | LMNII | PMNII | GC | Fibrosis | Angiogenesis |
|---|---|---|---|---|---|---|---|
| 3 | 1.3 | + | 2 | Eos 1 | 0.7 | 2 | 5.6 |
| 7 | 2.9 | + | 1 | Eos 1 | 0.6 | 2 | 1.7 |
| 9 | 5.0 | + | 1 | 0 | 32.0 | 2 | 3.7 |
| 11 | 2.8 | + | 1 | 0 | 6.2 | 1 | 6.3 |
| 12 | 3.0 | + | 1 | 0 | 27.0 | 1 | 5.9 |
LMNII, lymphomononuclear inflammatory infiltrate (0-3); PMNII, polymorphonuclear inflammatory infiltrate (0-3), Eos, cellular predominance of eosinophils, GC (giant cells), mean number of giant cells per high-power field (40 ×); Fibrosis, intensity of fibrosis observed; Polymer, presence or absence of the biopolymer; Angiogenesis, mean number of vascular spaces per high-power field (40 ×); thickness, measurement of the vocal fold thickness at its point of greatest width.
Histological characteristics found in Group II - 90 days - Control vocal fold.
| Rabbit | Thickness | Polymer | LMNII | PMNII | GC | Fibrosis | Angiogenesis |
|---|---|---|---|---|---|---|---|
| 3 | 1.2 | 0 | 0 | 0 | 0 | 0 | 0 |
| 7 | 2.0 | 0 | 0 | 0 | 0 | 0 | 0 |
| 9 | 1.9 | 0 | 0 | 0 | 0 | 0 | 0 |
| 11 | 1.4 | 0 | 0 | 0 | 0 | 0 | 0 |
| 12 | 1.2 | 0 | 0 | 0 | 0 | 0 | 0 |
LMNII, lymphomononuclear inflammatory infiltrate (0-3); PMNII, polymorphonuclear inflammatory infiltrate (0-3), Eos, cellular predominance of eosinophils, GC (giant cells), mean number of giant cells per high-power field (40 ×); Fibrosis, intensity of fibrosis observed; Polymer, presence or absence of the biopolymer; Angiogenesis, mean number of vascular spaces per high-power field (40 ×); thickness, measurement of the vocal fold thickness at its point of greatest width.
All rabbits showed the presence of SCB. No specimen exhibited rejection of the material, necrosis or calcification. The vocal folds injected with saline solution showed no inflammatory reaction in both groups. In rabbits number 2, 3, 4 and 7, the presence of polymorphonuclear inflammatory infiltrate consisted predominantly of eosinophils.
When comparing group I (21 days) and group II (90 days) regarding the presence of fibrosis in the vocal folds with biopolymer, lymphomononuclear inflammatory infiltrate, polymorphonuclear inflammatory infiltrate and the analysis between the predominance of giant cells and angiogenesis between the biopolymer injection groups, there was no statistically significant difference between groups.
Regarding differences in thickness between the implanted and control vocal folds,the difference was statistically significant, as shown in Table 5.
Comparative analysis of thickness, measured at its greatest width in millimeters, of rabbit vocal folds injected with sugarcane biopolymer and control vocal folds.
| Rabbit (n) | SCB/Thickness (mm) | Control/Thickness (mm) | |
|---|---|---|---|
| 1 | 2.2 | 1.3 | p=0.0058 |
| 2 | 3.1 | 2.9 | |
| 4 | 4.0 | 5.0 | |
| 5 | 4.0 | 2.8 | |
| 10 | 2.2 | 3.0 | |
| 3 | 1.3 | 1.2 | |
| 7 | 2.9 | 2.0 | |
| 9 | 5.0 | 1.9 | |
| 11 | 2.8 | 1.4 | |
| 12 | 3.0 | 1.2 |
Group I, 21 days and Group II, 90 days. Significant for comparison. (Mann-Whitney U, WilcoxonTest).
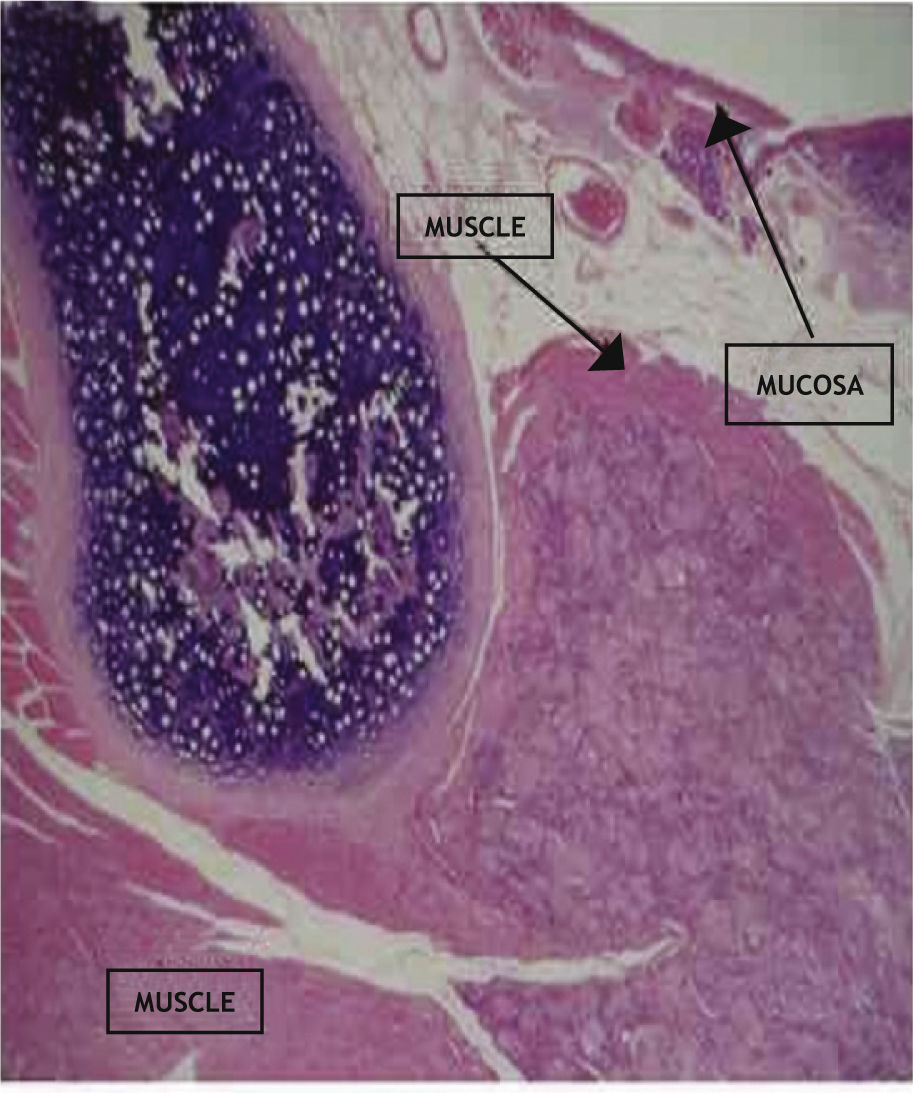
There were no epithelial layer lesions in the vocal folds evaluated by microscopy, as shown in Fig. 2.
DiscussionVocal fold alterations that involve a reduction of volume and failure of glottal closure result in an exaggerated air escape during speech, which makes the voice excessively breathy.
For some of these cases, surgical procedures resulting in an increase of the affected vocal fold volume to reduce the glottal gap may be indicated. However, placing a material inside the fold can alter its vibrational properties, either by the presence of the material itself, or by the inflammatory changes it will produce in the subepithelial space, resulting in voice hoarseness.
To date, there has been no consensus in the literature regarding the type of material to be used in vocal folds. Several materials have been tested, including fat, which seems to be the most used in Brazil, mainly due to its prompt availability and low cost and that can be inserted through a submucosal pouch in the vocal fold under general anesthesia.10
Others, such as hydroxyapatite have had positive literature references but are expensive. One of the advantages of injectable substances is that they can be applied without the need for general anesthesia, but this is not the case for hydroxyapatite, fascia and areolar tissue, which need to be introduced after the creation of a submucosal pouch in the vocal fold.5,6,11,16–18
The sugarcane biopolymer has been the subject of numerous studies. Several studies have assessed its properties in different systems, such as the restoration of vascular and abdominal walls, as well as the middle ear inflammatory response.9–15
The use of the SCB gel on the vocal folds has never been described. This material was chosen because it is injectable and meets certain criteria, such as the fact that it is easy to handle, its biocompatibility, low cost and low probability of infection, which makes it a good candidate for correction of pathological vocal fold alterations.6,18
In our study, we found a mild to moderate inflammatory response in both temporal groups of vocal folds with SCB. There was no significant inflammatory reaction caused by the surgical procedure in the control vocal folds. Other substances studied in the literature showed similar results in relation to the inflammatory response between the cases and their controls. Duprat, in 20015 studied the behavior of fat grafts in rabbits and obtained similar inflammatory responses between cases and controls. The author states that the fat did not appear to interfere with the inflammatory response in the surgical wound of the control vocal fold. However, the surgery performed by this author is different than what we did. He created a pouch to receive the fat graft in the vocal fold and the control was the similar surgical trauma without graft placement. Autologous fat obviously has a lower cost than the SCB.18
The trauma of saline solution injection was not sufficient to trigger inflammatory activity or a fibrogenic responses in our observations and in the literature using a similar methodology. In 2007, Perazzo et al.19 used a surgical technique similar to ours to inject hyaluronic acid in vocal folds of rabbits. This author obtained similar results to those of our study in control vocal folds, in which the injection of saline solution was used. We observed virtually no inflammatory response in the vocal folds injected with saline solution. Hyaluronic acid, as well as the SCB, can be injected under local anesthesia in humans, but it has a higher cost.
The use of SCB in gel form resulted in greater inflammatory responses than their controls and this inflammatory activity persisted throughout the study period. The 3-week and 3-month cases were statistically equivalent for all inflammatory variables studied. The inflammatory response basically consisted of lymphomononuclear infiltrate of mild to moderate intensity and some rabbits showed polymorphonuclear infiltrate with eosinophil predominance.
This finding was not described in other studies performed with other substances. There has been no description of the presence of eosinophils in literature with SCB use elsewhere. These cells are responsible for allergic reactions and their presence should be further studied, as the possibility of contaminating substances such as dust used in surgical gloves may have contributed to their appearance, as well as the fact that the infiltration site or gel formulation may be related to this finding.
The gel SCB-induced fibrogenesis was mild to moderate in intensity. The observed pattern showed a peripheral predominance with apparent formation of fibrous capsules. Some specimens, such as rabbit 2 vocal fold, with SCB, showed a nodular and non-homogeneous pattern. Even in cases where the biopolymer location was more superficial, there was no alterations in the epithelial layer or Reinke space. As reported in the works of Flinck C,20 Thibeault SL21,22 and Longaker MT23 the preservation of the subepithelial layers is extremely important to preserve the mucosal wave and voice quality.
This fibrogenic response with absence of epithelial repercussions – no retractions, no pus formation or extrusion – suggests that the gel biopolymer can be bioactive and preserve normal epithelial vibratory function. However, the presence of an inflammatory process in the subepithelial layers may suggest a change in the physical properties of Reinke space, which also has major importance in voice production. Any reduction in the amplitude of the mucosal wave can cause voice hoarseness. However, this inflammatory response also occurred with all the other materials studied before.
Although there are foreign body giant cells, we observed elements that suggested good integration of the biopolymer with tissues, such as the constant presence of angiogenesis and fibrous cords that passed through the biopolymer. Because of the SCB bio-integration, with cells invading its intimacy, it was not possible to easily identify in the studied slides the precise limits of the material within the vocal fold and sometimes the biopolymer seemed to be disseminated into the tissue, which prevented the measurement of total area occupied by the SCB. For this reason, a more detailed analysis of the degree of biopolymer absorption has not been possible. However, sinceit was more difficult to determine the biopolymer region in the 3-month group, there may be some biopolymer absorption over time, even if it was partial.
On the other hand, we analyzed vocal fold thickness in its greatest cross-sectional dimension with the purpose of assessing the increase in volume. The larynges were not sectioned according to a standard in all rabbits, and so the comparison between temporal groups (21 days and 90 days) would have a significant bias. However, as the vocal cords of the each larynx were sectioned in the same way, it was possible to compare the thickness of those that received SCB with the control ones.
The vocal fold increase was significant and this is extremely important, as one of the main objectives of biomaterial injection in the vocal folds is precisely to increase its volume and the consequent glottal closure, allowing improved vocal production.
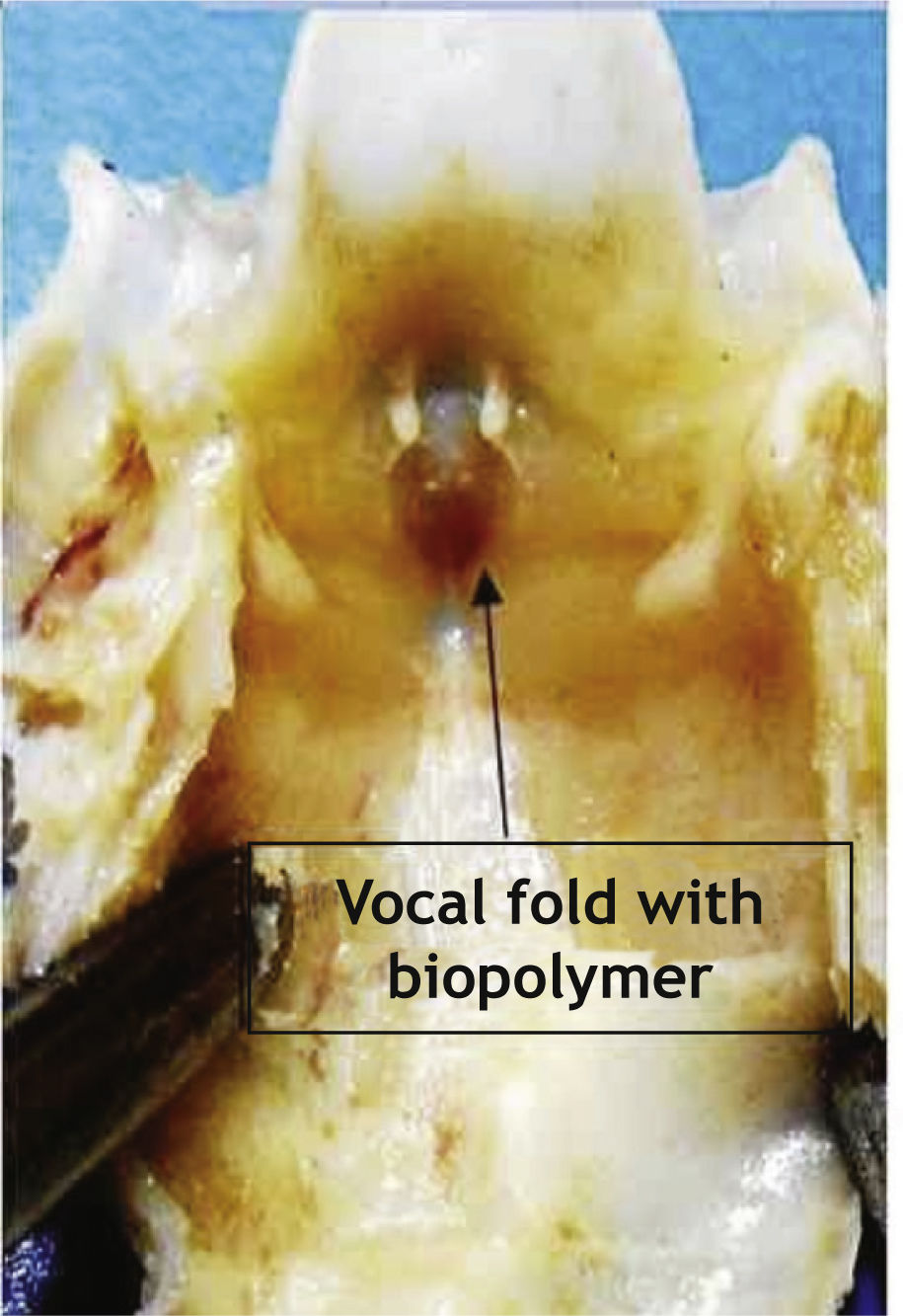
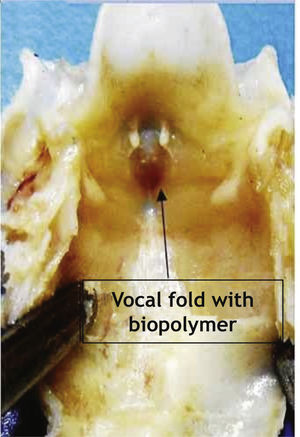
In contrast to what occurred in cases that received other substances, there were no cases of biopolymer rejection or complete absorbtion in the present study. The SCB remained present in all the injected vocal folds. During the macroscopic analysis, the vocal fold with SCB was sometimes easily identified by the difference in volume caused by the presence of the biopolymer (Fig. 3).
ConclusionDespite causing an inflammatory reaction in vocal fold tissues that persisted throughout the study period, injected SCB remained in the vocal fold after three months considerably increasing vocal fold volume for up to 90 days in.
Conflicts of interestThe authors declare no conflicts of interest.
Please cite this article as: Leão RAS, Assis RC, Caldas Neto SS, Lira MMM, Vasconcelos SJ. Effect os sugarcane biopolymer gel injected in rabbit vocal fold. Braz J Otorhinolaryngol. 2014;80:220-5.